PROPERTIES OF MATTER
| Site: | Newgate University Minna - Elearning Platform |
| Course: | General Physics I |
| Book: | PROPERTIES OF MATTER |
| Printed by: | Guest user |
| Date: | Monday, 6 April 2026, 2:18 PM |
Description
The Properties of Matter course explores the fundamental characteristics that define and distinguish materials, focusing on both physical and mechanical attributes. Key concepts include density, which explains why oil floats on water; pressure, as seen in how sharp knives cut more effectively due to higher pressure over smaller areas; and elasticity, which is crucial in designing springs and shock absorbers. The course also covers floatation using Archimedes’ Principle, which helps explain how ships float despite their massive weight. Additionally, it examines surface tension, observable when insects like mosquitoes walk on water, and viscosity, which influences the flow of engine oils in automobiles.
1. INTRODUCTION
1.0 INTRODUCTION
Properties of matter are the characteristics or attributes of a substance that help define and distinguish it from other substances. These properties can be observed or measured and are classified into physical properties (which do not change the substance’s identity) and chemical properties (which describe how a substance interacts with other substances and changes composition).
1.1 Density
1.1.1 Density is a property of matter. It tells us about how concentrated the matter is in a particular material. Density is a constant for a given material under specific conditions. Density is defined as the mass per unit volume of a substance.
Density = mass/volume ρ = m/v
The symbol used here for density, ρ, is the Greek letter rho.
The standard unit for density in the SI system is kgm−3, but you may also find values quoted in gcm−3.
It is useful to remember that these units are related by: 1000 kgm−3 = 1 gcm−3 and that the density of water is approximately 1000 kgm−3.
1.1.2 Mass: is the measurement of the amount of matter in an object. It is typically measured in kilograms (kg) or grams (g) and remains constant regardless of location, unlike weight, which depends on gravity.
Volume is the amount of space that a substance or object takes. It is measured in units such as liters (L), cubic meters (m³), cubic centimeters (cm³), or milliliters (mL)
1.1.3 Examples
1. A cube of copper has a mass of 240g. Each side of the cube is 3.0cm long. Calculate the density of cop per in gcm−3 and in kgm−3. (Surface Area)
2. The density of steel is 7850kgm−3. Calculate the mass of a steel sphere of radius 0.15m.
2. PRESSURE
1.2 Pressure
A fluid (liquid or gas) exerts pressure on the walls of its container, or on any surface with which it is in contact. A big force on a small area produces a high-pressure
Pressure is defined as the normal force acting per unit cross-sectional area.
pressure = normal force/cross-sectional area p = F/A
Force is measured in newtons and area is measured in square metres. The units of pressure are thus newtons per square metre (Nm−2), which are given the special name of pascals (Pa).
1Pa = 1Nm−2
1.2.1 Examples
1. A chair stands on four feet, each of area 10cm2. The chair weighs 80N. Calculate
the pressure it exerts on the floor.
2. A rectangular water tank of weight 4.5 x 103N measures 2.0m by 1.5m by 1.2m. Calculate the minimum pressure it can exert when resting on a horizontal surface.
3. A rectangular block of dimensions 2.0mxl.0mx0.5m weighs 200N. Calculate the maximum pressure exerted by the block on a horizontal floor.
4. A rectangular tank contains water to a depth of 2m. If the base is 4m x 3m calculate the force on the base. (Density of water = 103kgm3, g=10ms-2)
5. The pressure exerted on the floor by a boy standing on 4 Feet. If the weight of the boy is 40kg. Assuming each shoe's contact area is 6cm by 25cm (Take 1kg wt = 10N).
3. PRESSURE IN FLUID
1.3 Pressure in a fluid
Pressure in fluids (liquids and Gases) increases with depth. This is because liquids with greater density have greater weight acting downwards. The pressure of the liquids also increases with their density. For example, Divers, Airplanes, and Mountain climbers experience varied pressures as they move below or above the Earth's surface.
As the surface increases in height above the ground level, the number of air molecules above the surface decreases. The pressure of the atmosphere is caused by the air molecules colliding with the surface of the Earth. Hence, the atmosphere gets less dense as we increase in altitude (move away from the surface of the Earth). At the Earth's surface, there are a greater number of molecules in a given volume, i.e., the Atmosphere is denser.
Atmosphere refers to the thin layer of air around the Earth's surface.
1.3.1 The pressure in a fluid depends on three factors:
The depth h below the surface
The density ρ of the fluid
The acceleration due to gravity, g
p = ρgh
In addition, since fluid is a liquid or gas its pressure applies in all directions. Fluids can be enclosed in a container or due to gravity.
1.3.2 Gravity: The weight of a fluid can exert pressure on anything underneath or below it and the relative movement of liquids or gas can supply pressure at the surface of the object. e.g water in a container, lake water, etc.
1.3.3. Air and Water Pressure Apply in all Directions
Now what is different about pressure caused by air or water is that the pressure pushing downward in a specific direction is equal to the pressure pushing upward and also at the other sides. For instance, pressure is equal in liquids at some point because when you submerge a square container or any solid shape in a liquid, the pressure is applied to all the sides (length, height, and width). Other relative examples are:
a. Swimming underwater: when you swim underwater, the pressure of the water gets on your body, the deeper you move down the greater the pressure.
b. Wind: the movement of fluids such as wind can apply pressure to an object in a way proportional to the surface area perpendicular to the direction of motion.
1.3.4 Examples
1. Calculate the pressure of water on the bottom of a swimming pool if the depth of water in the pool varies between 0.8m and 2.4m. (Density of water = 1000kgm−3.) If atmospheric pressure is
1.01 × 105Pa, calculate the maximum total pressure at the bottom of the swimming pool.
2. Estimate the height of the atmosphere if atmospheric density at the Earth’s surface is 1.29kgm−3. (Atmospheric pressure = 101kPa.)
3. A reservoir is filled with a liquid of density 2000kgm3. Calculate the depth at which the pressure in the liquid will be equal to 9100Nm2 (g=10m/s2).
4. A 5m x 4m x 3m vessel of negligible weight is filled with a liquid of density 2500kgm-3. If the vessel is placed on a flat surface, what is the maximum pressure it can exert?
4. ELASTICITY AND RELATED CONCEPT
1.3 Elasticity and Related Concepts
1. Elasticity:
Is the
ability of a solid material to return to its original shape and size after the
removal of external forces that caused its deformation. This property allows
materials to resist permanent shape changes and recover their initial form when
the applied stress is removed.
2. Deforming
Forces: These
forces alter a body's shape, size, or configuration by causing deformation.
3. Restoring Force: A restoring force
is an internal force exerted by a deformed body that acts in the opposite
direction of deformation, striving to return the body to its equilibrium state.
4. Elastic
Body: An
elastic body is one that regains its original shape and dimensions after the
removal of deforming forces. Common examples include a soccer ball, rubber
band, and spring.
1.4.1 Stress
When a deforming force acts perpendicular (normal) to the surface of a body, it
generates normal stress. This type of stress can be further classified
into:
When a deforming force acts parallel (tangentially) to the surface of a body,
it produces tangential stress, also known as shear stress. This
type of stress leads to a change in the shape of the body without altering its
volume.
1.4.3 Strain
Longitudinal Strain:
If the deforming force produces a change in length only, the strain produced
is called longitudinal or tensile strain.
It is defined as the ratio of change
in length to the original length.
Stress is defined as the restoring force per unit area of a material.
Stress = F/A
Unit of stress is N/m2 or Pascal Stress is of two types:
1.4.2 Types of Stress
1. Normal Stress:
a. Tensile Stress (when the force pulls the body apart, increasing its length)
·b. Compressive Stress (when the force pushes the body inward, reducing its length)
2. Tangential Stress (Shear Stress):
Strain is defined as the ratio of change in configuration to the original configuration, when a deforming force is applied to a body.
Strain is of three types:
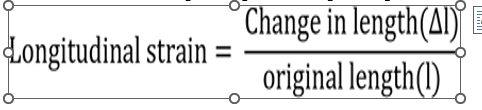
i. Longitudinal Strain: If the deforming force produces a change in length only, the strain produced is called longitudinal or tensile strain. It is defined as the ratio of change in length to the original length.
ii. Volumetric
strain: It is defined as the ratio of the change in
volume to the original volume
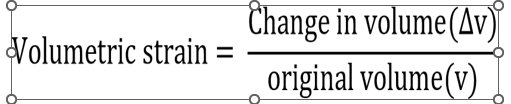
iii. Shearing strain: It is defined as the ratio of lateral displacement of a surface under the tangential force to the perpendicular distance between surfaces.
Strain is a ratio of two similar physical quantities, it has no units and dimensions.

5. HOOK'S LAW
5.1 Hooke’s law
Hooke’s law states that provided the elastic limit of an elastic material is not exceeded, the extension e, of the material is directly proportional to the applied force, F.
Thus, 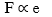
F = ke
where k is a constant of proportionality called elastic constant or force constant of the material. If F is in newtons and e is n metres, the elastic constant is given by
k = F/e (Nm-1)
where k is a constant of proportionality called elastic constant or force constant of the material. If F is in newtons and e is n metres, the elastic constant is given by
k = F/e (Nm-1)
1.5.1 Elastic constant of an elastic material is the force required to give unit extension
a. Young’s modulus
Young’s modulus (Y) is defined as the ratio of normal stress to the longitudinal strain.
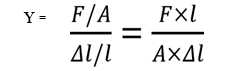
Since strain is a dimensionless quantity, the unit of Young’s modulus is the same as that of stress i.e., Nm–2 or Pascal (Pa).
b. Bulk’s Modulus
Bulk’s modulus (B) is defined as the ratio of normal (hydraulic) stress to the volumetric strain.
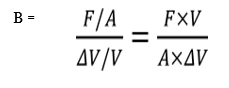
SI unit of bulk modulus is the same as that of pressure i.e., Nm–2or Pa
c. Shear modulus
The ratio of shearing stress to the corresponding shearing strain is called the shear modulus of the material and is represented by. It is also called the modulus of rigidity.
G= Tangential stress /Shear strain
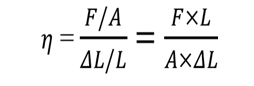
SI unit of shear modulus is Nm–2or Pa.
1.5.2 Examples
1. An object weighing 0.6N is hung on a spiral spring and causes it to extend by 6.0cm. The object is removed and a block of wood replacing it causes the spring to extend by 10.0cm. What is the weight of the block of wood?
2. A force of 40N applied at the end of a wire of length 4m and diameter 2.00mm process and extension of 0.24mm. calculate;
(a) Stress on the wire
(b) strain in the wire
(c) Young’s Modulus for the material of the wire (π=3.14).
3. A load of 5N gives an extension of 0.56cm in a wire which obeys Hooke’s law. What is the extension caused by a load of 20N?
4. A force of magnitude 500N is applied to the free end of a spiral of force constant 1.0 x 104Nm-1. Calculate the energy stored in the stretched spring.
6. FLOATATION
1.6 Floatation
Floatation is the ability of an object to float or rise to the surface a fluid it is immersed in.
1.6.1 Buoyancy
Buoyancy is a familiar phenomenon: A body immersed in water seems to weigh less than when it is in air. When the body is less dense than the fluid, it floats. The human body usually floats in water, and a helium-filled balloon floats in air.
7. ARCHIMEDE'S PRINCIPLE
1.7 Archimedes’ Principle
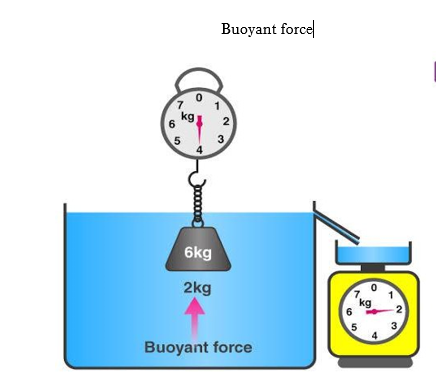
Archimedes’ principle states that when a body is completely or partially immersed in a fluid, the fluid exerts an upward force on the body equal to the weight of the fluid displaced by the body.
1.7.1 Upthrust
Any object placed in a fluid such as water or air experiences an upwards force. This is what makes it possible for something to float in water. Upthrust arises from the pressure which a fluid exerts on an object. The deeper you go, the greater the pressure. So there is more pressure on the lower surface of an object than on the upper surface, and this tends to push it upwards. If upthrust is greater than the object’s weight, it will float up to the surface
When a balloon floats in equilibrium in air, its weight (including the gas inside it) must be the same as the weight of the air displaced by the balloon.
A fish’s flesh is denser than water, yet a fish can float while submerged because it has a gas-filled cavity within its body. This makes the fish’s average density the same as water’s, so its net weight is the same as the weight of the water it displaces.
A body whose average density is less than that of a liquid can float partially submerged at the free upper surface of the liquid. The greater the density of the liquid, the less of the body is submerged. When you swim in seawater density your body floats higher than in fresh water (1000 kg/m3).
8. SURFACE TENSION
1.8 Surface Tension
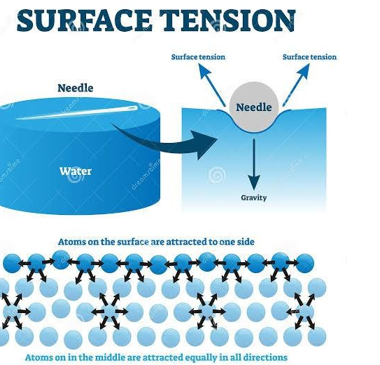
An object less dense than water, such as an air-filled beach ball, floats with part of its volume below the surface. Conversely, a paper clip can rest atop a water surface even though its density is several times that of water. This is an example of surface tension.
Surface Tension is the property of a liquid due to which its free surface behaves like stretched membrane and acquires minimum surface area. It is given by force per unit length.
T = F/l SI unit is N/m
1.8.1 Cohesive force
This is the force of attraction between molecules of the same substance. For instance the attraction between water molecules.
1.8.2 Adhesive force
This is defined as the force that binds molecules of different substances together. For example the attraction between water and air molecules.

Surface tension on a needle suspended on the surface of water
1.8.3 Effect of heat on surface tension
In general, surface tension decreases when temperature increases and vice versa. This is because cohesive forces decrease with an increase of molecular thermal activity. The influence of the surrounding environment is due to the adhesive action liquid molecules have at the interface.
9. VISCOUSITY
1.8 Viscosity
Viscosity is the property of liquid due to which it opposes the relative motion between the layers of fluid. It is also known as liquid friction.
SI unit of viscosity is Pascal-second (Pa.s) and CGS unit is Poise.
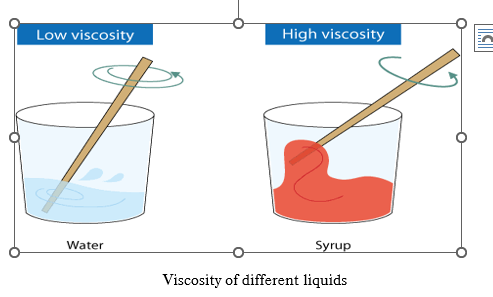
1.8.1 Effect of heat on viscosity
In liquids the source for Viscosity is considered to be atomic bonding. As we understand that, with the increase of temperature, the bonds break and make the molecule free to move. So, we can conclude that the viscosity decreases as the temperature increases and vice versa.
In gases, due to the lack of cohesion, the source of viscosity is the collision of molecules. Here, as the temperature increases the viscosity increases and vice versa. This is because the gas molecules utilize the given thermal energy in increasing its kinetic energy that makes them random and therefore resulting in more the number of collisions.