PROPERTIES OF MATTER
The Properties of Matter course explores the fundamental characteristics that define and distinguish materials, focusing on both physical and mechanical attributes. Key concepts include density, which explains why oil floats on water; pressure, as seen in how sharp knives cut more effectively due to higher pressure over smaller areas; and elasticity, which is crucial in designing springs and shock absorbers. The course also covers floatation using Archimedes’ Principle, which helps explain how ships float despite their massive weight. Additionally, it examines surface tension, observable when insects like mosquitoes walk on water, and viscosity, which influences the flow of engine oils in automobiles.
8. SURFACE TENSION
1.8 Surface Tension
An object less dense than water, such as an air-filled beach ball, floats with part of its volume below the surface. Conversely, a paper clip can rest atop a water surface even though its density is several times that of water. This is an example of surface tension.
Surface Tension is the property of a liquid due to which its free surface behaves like stretched membrane and acquires minimum surface area. It is given by force per unit length.
T = F/l SI unit is N/m
1.8.1 Cohesive force
This is the force of attraction between molecules of the same substance. For instance the attraction between water molecules.
1.8.2 Adhesive force
This is defined as the force that binds molecules of different substances together. For example the attraction between water and air molecules.

Surface tension on a needle suspended on the surface of water
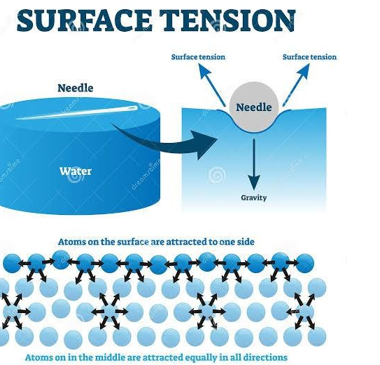
1.8.3 Effect of heat on surface tension
In general, surface tension decreases when temperature increases and vice versa. This is because cohesive forces decrease with an increase of molecular thermal activity. The influence of the surrounding environment is due to the adhesive action liquid molecules have at the interface.

