PROPERTIES OF MATTER
The Properties of Matter course explores the fundamental characteristics that define and distinguish materials, focusing on both physical and mechanical attributes. Key concepts include density, which explains why oil floats on water; pressure, as seen in how sharp knives cut more effectively due to higher pressure over smaller areas; and elasticity, which is crucial in designing springs and shock absorbers. The course also covers floatation using Archimedes’ Principle, which helps explain how ships float despite their massive weight. Additionally, it examines surface tension, observable when insects like mosquitoes walk on water, and viscosity, which influences the flow of engine oils in automobiles.
9. VISCOUSITY
1.8 Viscosity
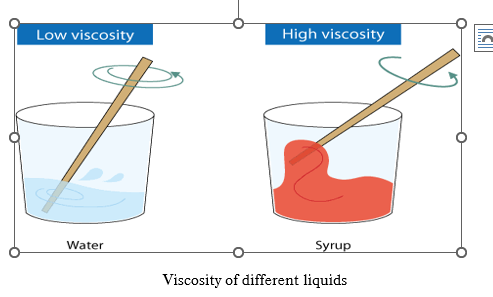
Viscosity is the property of liquid due to which it opposes the relative motion between the layers of fluid. It is also known as liquid friction.
SI unit of viscosity is Pascal-second (Pa.s) and CGS unit is Poise.
1.8.1 Effect of heat on viscosity
In liquids the source for Viscosity is considered to be atomic bonding. As we understand that, with the increase of temperature, the bonds break and make the molecule free to move. So, we can conclude that the viscosity decreases as the temperature increases and vice versa.
In gases, due to the lack of cohesion, the source of viscosity is the collision of molecules. Here, as the temperature increases the viscosity increases and vice versa. This is because the gas molecules utilize the given thermal energy in increasing its kinetic energy that makes them random and therefore resulting in more the number of collisions.

